I want to begin with a reminder that often gets lost in public discussions: the Covid mRNA vaccines are genuinely novel medical products.
Prior to the emergency authorizations in 2020, mRNA vaccine technology had never been deployed at scale in humans. Only two clinical trials, one from Pfizer-BioNTech and one from Moderna, had ever tested this platform in people. Altogether, roughly 37,000 individuals had ever received an mRNA vaccine in the history of medicine (not including the earlier experience with rabies, CMV, and cancer vaccines limited to much smaller early-phase studies). That is not a criticism; it is simply a statement of fact. But it does mean that the long-term safety profile of these products was, and remains, incompletely understood.
What follows is familiar to nearly all molecular biologists. It is complicated but I try to simplify it given the stakes. It’s important to clearly lay out the molecular framework for everyone because how these vaccines are made directly determines what is inside the vial. And what is inside the vial, once injected, will travel throughout the body and activate a cascade of events that can lead to long-term health implications.
In Vitro Transcription Is Not Just a Manufacturing Detail
Modified mRNA vaccines are produced using a process called in vitro transcription (IVT). IVT is the method used to synthesize the modified mRNA that ultimately becomes the active ingredient in the vaccine.
This is not a trivial technicality. IVT fundamentally shapes the molecular composition of the final product.
Scientists at BioNTech, including those directly involved in developing the Pfizer vaccine, have published a detailed review1 describing how IVT reactions generate not only the intended full-length mRNA, but also a range of byproducts and impurities, how these are typically removed, and what their biological consequences may be if they persist. These manufacturing directions along with the byproducts they create were also described in detail by Moderna in their patents (US10,653,712 B2 and US10,077,439 B2). But more importantly, this molecular biology was well established long before Covid. None of this is speculative.
The Starting Material: DNA Templates
At its core, an IVT reaction begins with double-stranded DNA that encodes the desired protein. In this case, the SARS-CoV-2 spike protein.
The spike-encoding sequence used in mRNA vaccines is genetically modified to improve stability and cellular tolerance, including two amino-acid substitutions that make it distinct from the viral spike. That modification is intentional.
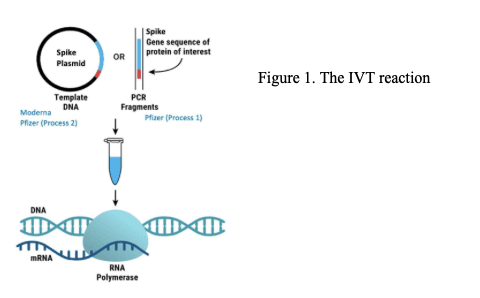
The DNA template itself can take different forms. During Pfizer’s early clinical trials, PCR-generated DNA fragments were used. However, the commercial manufacturing process relied on DNA derived from plasmids. This matters because plasmids contain additional regulatory sequences. In Pfizer’s case, these include elements such as SV40 promoter and ori sequences, which raise concerns if they were to enter human cells.
Once this DNA template is added to the IVT reaction, along with RNA polymerase and other components, it is transcribed into mRNA (Figure 1).

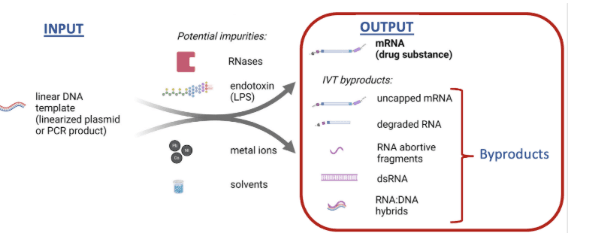
While the desired output of IVT is the intended full-length mRNA product, the actual output is more complex. These include various byproducts in the form of (1) various RNA species including double-stranded RNA (dsRNA), (2) DNA attached to RNA (RNA–DNA hybrids), and (3) the free DNA from the original template (Figure 2).
The formation of these byproducts is well-documented and unavoidable and is why downstream purification is absolutely essential for safety.

Purification Has Known Limitations
After manufacturing there are two purification steps needed to first remove the DNA and then the remove the RNA byproducts (Figure 3):
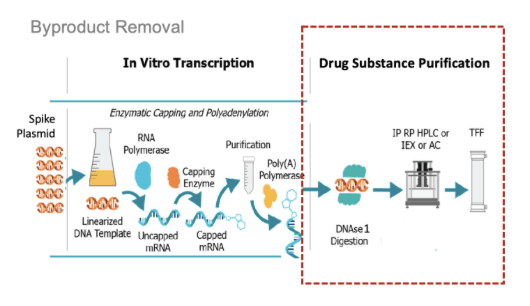

To remove the DNA, an enzyme is added to the reaction mixture called DNase I, which is commonly used to degrade contaminating DNA. While DNase I is effective against the free template DNA, multiple studies, including work by BioNTech scientists themselves, show that DNase I is inefficient at removing the DNA attached to RNA (RNA–DNA hybrids).
This limitation is not controversial. It is documented in the literature.
What Independent Analyses Have Shown
This context is crucial for interpreting recent independent analyses of finished vaccine vials.
Researchers3 and regulators4 have reported detecting DNA contaminants in essentially every vial tested. These contaminants included both double-stranded DNA and RNA–DNA hybrids that appeared resistant to DNase I digestion.
In some samples, spike-encoding DNA was present at levels more than 100-fold higher than other plasmid sequences5, suggesting uneven or incomplete digestion. Sequencing and quantitative PCR analyses further detected DNA fragments averaging ~200 base pairs in length, with some exceeding 4 kilobases. In several cases, sequences spanning nearly the entire plasmid were observed.
Taken together, these findings raise serious questions about the consistency and completeness of purification during large-scale manufacturing, and about the potential biological consequences of residual nucleic acids in people.
Why Nucleic Acid Contaminants Matter Biologically
RNA and DNA are potent activators of innate immune pathways. This is not speculative. Pattern-recognition receptors and the cGAS-STING pathway respond robustly to foreign nucleic acids, triggering inflammation, growth inhibition, and even cell death.
These mechanisms are precisely why gene-therapy products are subject to stringent safety oversight.
Ironically, the Covid mRNA vaccines were designed with modifications specifically to reduce this potent innate immune activation. But RNA–DNA hybrids and DNA fragments will still provoke strong immune responses despite those modifications.
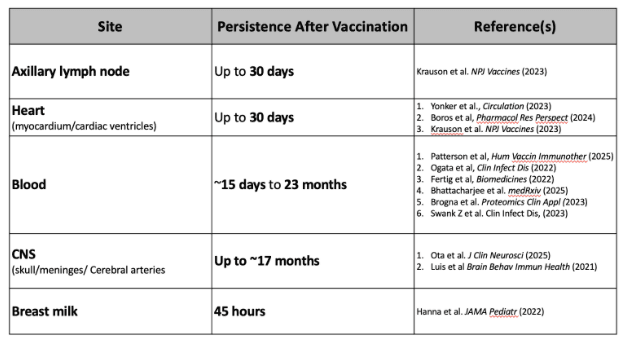
Persistence Raises New Questions
There is now substantial evidence showing that spike mRNA and protein persists in human tissues for weeks, months, and even years after vaccination (Table 1).
We do not yet know whether this persistence reflects prolonged mRNA stability, continued translation, or DNA-based mechanisms. But given the plausibility of DNA integration, and long-lived non-integrated plasmid DNA in muscle cells,6 it is not unreasonable to assume the persistence of Spike mRNA, protein, and antibodies against Spike years after vaccination is not unrelated to the DNA impurities and byproducts following IVT.

Short- and Long-Term Safety Implications
Taken together, these data raise several important safety considerations.
First, acute immune reactions, including cytokine storms and anaphylaxis, have been reported immediately following vaccination. Such strong inflammatory responses should not be dismissed outright as unrelated to impurities, particularly given what is known about nucleic-acid-induced immune activation.
Second, and more critically, are long-term risks. Persistent spike expression could plausibly contribute to chronic immune syndromes. More concerning still is the possibility of DNA integration, which carries risks of insertional mutagenesis or gene disruption. This means a risk for cancer or developmental defects depending on where and at what age the DNA was integrated.
Notably, the FDA itself states in its information sheets that these vaccines have not been evaluated for carcinogenicity (cancer formation) or genotoxicity (DNA damage), a point that would be routine and expected in gene-therapy oversight, where long-term monitoring is standard.
The Regulatory Gap around DNA in mRNA Vaccines
Since there really is no longer any dispute that residual DNA exists in mRNA vaccines, the issue is whether current guidelines and safety limits are appropriate for mRNA vaccines. We’ve been assured that the DNA byproducts are within the limits set forth in the regulatory guidelines. So what is the FDA guidance surrounding DNA byproducts and contaminants?
The most commonly cited FDA guidance on residual DNA (≤10 ng per dose) was developed for viral vaccines produced in living cells that are fragmented and “naked,” with limited ability to enter human cells. However, mRNA vaccines are not produced in cells, their residual DNA is not host-cell derived, and most importantly, the DNA in the mRNA vaccines is not naked. It is associated with LNP delivery systems, which specifically make it very easy for the DNA to get inside cells. The FDA’s 2010 guidance is clear that it does not establish a relevant safety threshold for DNA associated with LNP-based products.
The other commonly cited guidance is from the WHO for recombinant protein therapeutics that address residual DNA in products such as monoclonal antibodies or hormones produced in engineered cells. Here again, the residual DNA originates from host cells or expression plasmids, is present as trace, non-encapsulated DNA (naked), and the final product is a purified protein, not a nucleic acid–based therapy (mRNA vaccine). So this guidance does not apply to mRNA vaccines.
Neither the FDA nor the WHO regulatory standards most often cited for residual DNA were developed for mRNA vaccines, and do not directly address this safety issue.
What the WHO Said About mRNA Vaccines — After Deployment
In 2022, the World Health Organization issued guidance specifically addressing mRNA vaccines7. Notably, this document was released after the global rollout of these products. It specifically states this guidance was in response to: “the safety, production and regulatory issues associated with this new technology.” The document also makes several important statements:
“Because detailed information is not yet available on the methods used for production, controls are not yet standardized for safe and efficacious mRNA vaccines, and certain details remain proprietary and thus not publicly available, it is not feasible to develop specific international guidelines or recommendations at this time.”
It further states: “The detailed production and control procedures…should be discussed with and approved by the NRA [National Regulatory Authority] on an individual case-by-case basis.”
The WHO acknowledges that controls for mRNA vaccines were not yet standardized and that it was not feasible to establish specific international guidelines or recommendations. Furthermore, regulatory oversight is required for case-by-case evaluation by national authorities.
This was stated after the mRNA vaccines had been deployed.
And as of the writing of this Substack, the FDA has still not established standardized guidelines for mRNA vaccines and provided any evidence and safety-based data to support any limits of DNA in mRNA vaccines.
Finally, it bears repeating: while mRNA technology is not new, before Covid, it was regulated as gene therapy, not as a traditional vaccine. The safety issues surrounding the DNA byproducts in Covid vaccines are going to be the same with any mRNA vaccine, including those for flu, RSV, or even mRNA vaccines for cancer.
This is because mRNA products are fundamentally different. They must enter cells and instruct them to produce a foreign protein. This is unlike any other conventional vaccines that deliver the protein directly. There is no clinical precedent for this platform, and there is no clinical precedent for repeated dosing. And certainly no precedent at population scale.
At this stage, with no pandemic, the accumulating mechanistic data and clinical observations, and the proliferation of mRNA vaccine products hitting the market, we need transparency and direct engagement with serious safety studies from regulators, particularly the FDA establishing critical guidelines for manufacturing these products – especially as it relates to DNA byproducts .
Novel technology demands novel scrutiny – not silence, gaslighting, or censorship.
References
1 https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2024.1426129/full
2 Webb C, Ip S, et al Mol Pharm. 2022 Apr 4;19(4):1047-1058. doi: 10.1021/
3 https://www.tandfonline.com/doi/10.1080/08916934.2025.2551517?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed
4 https://www.tga.gov.au/resources/publication/tga-laboratory-testing-reports/summary-report-residual-dna-and-endotoxin-covid-19-mrna-vaccines-conducted-tga-laboratories.
5 https://zenodo.org/records/17832183; https://www.scstatehouse.gov/CommitteeInfo/SenateMedicalAffairsCommittee/PandemicPreparedness/Phillip-Buckhaults-SC-Senate-09122023-final.pdf
6 Wang et al. (2004) – “Detection of integration of plasmid DNA into host genomic DNA following intramuscular injection and electroporation” (Gene Therapy, 2004). In mice, naked plasmid DNA was injected intramuscularly, followed by electroporation to enhance uptake. Using a highly sensitive PCR on purified genomic DNA (with gel separation to remove extrachromosomal forms), the authors identified four independent integration events at 4 weeks post-injection. Junction sequencing confirmed random integration sites (no preferential hotspots), consistent with non-homologous end joining. Integration frequency was low but measurable. This is one of the clearest demonstrations of actual spontaneous integration events in vivo for naked plasmid DNA in muscle. Of note – this study used enhanced DNA delivery via electroporation which one could compare to the enhanced delivery via LNPs.
Martin et al. (1999) – “Plasmid DNA Malaria Vaccine: The Potential for Genomic Integration after Intramuscular Injection” (Human Gene Therapy). This earlier study tested plasmid DNA IM in mice and used Southern blot hybridization and PCR on high-molecular-weight genomic DNA to probe for integration. While persistence was mostly extrachromosomal, they reported evidence suggestive of rare integration in some samples (though not as definitively sequenced as later works). It set a benchmark for low risk but acknowledged potential for very low-frequency events, influencing subsequent FDA guidance on DNA vaccines.
Ledwith et al. (2000) – “Plasmid DNA vaccines: Investigation of integration into host cellular DNA following intramuscular injection in mice” (Intervirology). Naked plasmid DNA injected IM in mice showed and while no detectable integration was observed, DNA was still detected in the quadriceps muscle up to 26 weeks. The DNA was extrachromosomal.
7 WHO Expert Committee on Biological Standardization 74th Report Annex 3. Evaluation of the quality, safety and efficacy of messenger RNA vaccines for the prevention of infectious diseases: regulatory considerations https://cdn.who.int/media/docs/default-source/biologicals/vaccine-standardization/annex-3—mrna-vaccines_who_trs_1039_web-2.pdf
Join the conversation:


Published under a Creative Commons Attribution 4.0 International License
For reprints, please set the canonical link back to the original Brownstone Institute Article and Author.