The CDC’s information page on Covid-19 vaccines contains the following bullet points on “How mRNA COVID-19 vaccines work:”
First, mRNA COVID-19 vaccines are given in the upper arm muscle or upper thigh, depending on the age of who is getting vaccinated.
After vaccination, the mRNA will enter the muscle cells. Once inside, they use the cells’ machinery to produce a harmless piece of what is called the spike protein…. After the protein piece is made, our cells break down the mRNA and remove it, leaving the body as waste.
Or, in other words, as we have long been told, “it” – the mRNA – “stays in the arm.” And then, after having instructed the muscle cells to produce the spike, is disposed of.
But look at the below picture from a recent presentation on mRNA vaccination at the European Parliament. The picture was posted on Twitter by Virginie Joron, a French member of the parliament. The speaker is no less an authority than Özlem Türeci, the Chief Medical Officer of BioNTech: the German biotech company that developed what has come to be known to most of the world as the “Pfizer” Covid-19 vaccine.

Have a closer look at Türeci’s slide, which tells a very different story than that which the CDC has been telling Americans for the last two years.
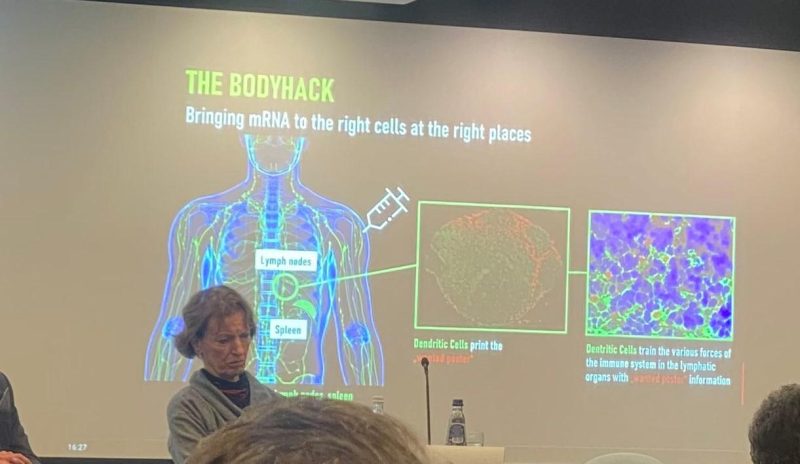
Far from “staying in the arm” and entering the muscle cells at the injection site, the injection site is only the point of departure for a journey that is supposed to take the mRNA rather to the lymph nodes. The subtitle of the slide is “Bringing mRNA to the right cells at the right places.” The deltoid is not the right place; the lymph nodes are.
Once in the lymph nodes, a specific sort of cell, the dendritic cells, is supposed to manufacture the spike protein: here colorfully described as the “wanted poster” that will help the immune system to identify the SARS-CoV-2 virus in case of subsequent exposure.
A passage from The Vaccine, the book that Türeci and her husband, BioNTech CEO Ugur Sahin, wrote with journalist Joe Miller, explains why BioNTech’s platform specifically targets the lymph nodes:
What Ugur learnt was that the location to which a vaccine delivers its ‘wanted poster’ really mattered. The reason for this, the couple’s team in Mainz later realised, was that not all dendritic cells … were created equal. The ones that resided in lymph nodes – of which the spleen is the largest – were particularly adept at capturing mRNA and making sure the instructions it carried were acted upon. These kidney-bean shaped organs, found under our armpits, in our groins, and at several other outposts in the body, are the information hubs of the immune system. (p. 98)
Indeed, Sahin and Türeci were so determined to get their mRNA into the lymph nodes that they had an earlier mRNA construct injected directly into the patient’s lymph nodes in the groin (p. 104).
Needless to say, such an approach was not likely to obtain wide acceptance as a vaccine! This is why the couple, as explained in their book, needed to package the mRNA in lipid nanoparticles, in order to ensure that mRNA administered by way of an intramuscular injection would, nonetheless, be widely distributed around the body and thus reach the lymph nodes.
This is to say that the wide biodistribution of the mRNA that came to light after rollout was never a bug. It is a feature of BioNTech’s mRNA technology. Having elicited an immune response by way of injection into the groin, Sahin is even said to have wondered, “How substantial could the immune response be if a vaccine got into all lymphatic tissues around the body, and recruited all the resident DCs [dendritic cells] into action?” (p. 105)
So, why has the CDC been lying about this for the last two years and insisting that the mRNA “stays in the arm?” Well, the obvious answer is that the idea of the mRNA staying at the injection site is reassuring, since otherwise we could fear systemic adverse effects of precisely the sort that have emerged since rollout.
It is worth noting, moreover, that in developing its vaccine, as discussed in my earlier article here, BioNTech simply skipped the so-called safety pharmacology studies whose purpose is precisely to test a candidate vaccine for potential systemic adverse effects – and regulators, including the FDA, let the company do it.
Published under a Creative Commons Attribution 4.0 International License
For reprints, please set the canonical link back to the original Brownstone Institute Article and Author.