Mandy Van Gorp was confident that her employer of 18 years, Eli Lilly and Company, would treat her fairly when she objected to its company-wide COVID-19 vaccine mandate. The pharmaceutical giant had promised to exempt employees with valid health or religious objections to the policy and she believed she had had both.
Despite presenting a doctor’s note in support of her exemption, citing an auto-immune disease, the company denied her request for a medical exemption. To add injury to the insult she felt, she tested positive for COVID-19 the day after receiving her rejection letter. She then appealed for a six-month deferral on grounds of the positive test. Lilly also denied that request. When she then raised her religious concerns, Lilly said she had missed the application deadline – a deadline that had lapsed several weeks before Lilly replied to her initial accommodation request.
The “toughest night was when we were sitting at the dinner table and my 12-year-old was sobbing, hysterically begging me to get the vaccine so I could keep my job,” recalled Van Gorp, a 42-year-old sales representative and mother of three. “I had to explain that my choice was not about money and that I felt God was leading me not to follow a mandate. It’s hard to explain that to a 12-year-old.”
Van Gorp’s experience was echoed by more than a dozen other former Lilly employees who recounted to RealClearInvestigations how the company’s vaccine mandate and its strict enforcement pushed them out.
Not only did they lose their jobs and health insurance, but some lost out on stock options and severance packages. Others struggled to collect unemployment, claiming Lilly misrepresented their dismissals to state offices. Salespeople who won exemptions said they too were effectively dismissed as the company pushed them toward roles in which they wouldn’t have direct contact with the public — jobs for which they often had little or no training and which would require them to relocate in some instances. When presented with a series of questions regarding its vaccine mandate policy and many of these claims, Lilly responded with a statement in support of vaccination as “guided by science.”
Lilly is just one of many major corporations that have publicly announced vaccine mandates for their employees. But specific policies have been imposed in private. The accounts of the former Lilly employees, including their never-before-disclosed allegations of unfair treatment, open a window on a largely secretive process that has roiled the American economy.
The objections of some also illuminate a trend seen across the healthcare industry: resistance to vaccines rooted in science and professional training, beyond objections solely based on religion or ideas of personal liberty. In this instance, those affected were in the business of manufacturing and selling drugs, including monoclonal antibodies used to treat COVID-19.
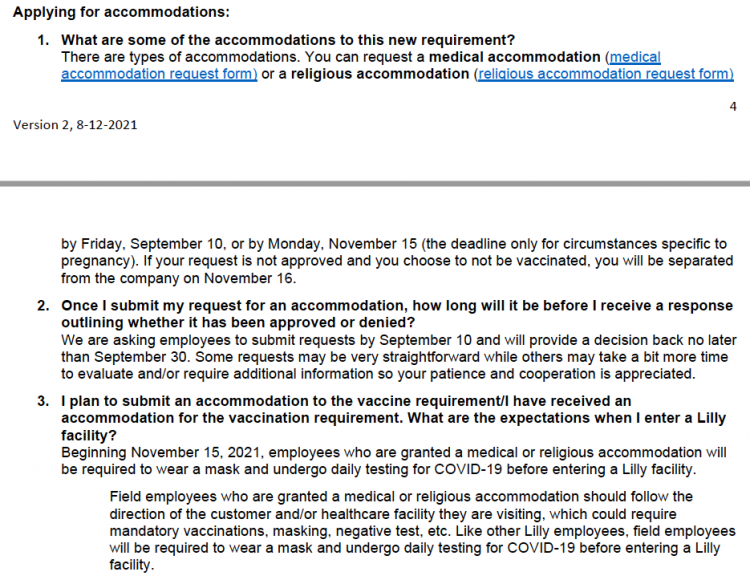
Lilly announced its vaccine mandate in August 2021, declaring that those “who do not meet this requirement or do not have an approved religious or medical accommodation in place by November 15th will be separated from the company.” The company had told its salespeople, who had worked remotely throughout the pandemic and then were allowed back in the field by March 2021, that those who received an exemption would remain secure in their jobs. They were instructed to “follow the direction of the customer and/or healthcare facility they are visiting, which could require mandatory vaccinations, masking, negative test, etc.” after Nov. 15th, as they had been doing since March.
While some employees approved of the mandate, others immediately pushed back. In a company-wide online forum discussion, the text of which RCI obtained, objectors raised various concerns ranging from the ethical – “What happened to individual liberty?” – to the scientific.
“Even though I’m vaccinated,” prefaced one participant, “I think that as a company who makes medicine and is fully aware of the amount of time it generally takes to get even non life-saving drugs tested and approved, this move makes no sense and goes against the safety and quality commitment Lilly tries to instill in its employees.”
Another employee questioned why the policy ignored evidence of protection provided by previous infection, writing: “The science of immunity from natural infection is being ignored, which is super disappointing considering we are a science-based company who developed an antibody treatment from those recovered.”
The employee was referring to the fact that Lilly had produced multiple monoclonal antibody treatments, which aim to neutralize active infections of COVID-19, as opposed to COVID-19 vaccines, which aim to protect patients from infection and severe illness.
Robin Clark, a former Lilly process engineer, was one employee who sought an exemption from Lilly. RCI reached Clark through a Telegram group of 85 ex-employees who have bonded over their shared outrage at how Lilly let them go. Unlike Clark, most members had been sales representatives. RCI’s investigation indicates the company may have subjected them to more onerous requirements in pursuing accommodations than non-customer-facing personnel.
Clark claims her opposition to vaccination was based on a sincere and long-held religious objection – one she did not want to disclose to her employer because “there’s a lot of discrimination against people who hold my beliefs.”
But Clark also had a pre-existing autoimmune disorder, so this was the basis on which she filed her initial exemption request.
In that request, she noted she had not received any vaccines since she was diagnosed with her condition in 1986, and quoted the Centers for Disease Control’s website, noting: “People with autoimmune conditions may receive a COVID-19 vaccine. However, they should be aware that no data are currently available on the safety of COVID-19 vaccines for people with autoimmune conditions.”
Clark also appended a letter from her doctor, provided to RCI, stating, “My medical professional assessment of this patient is that she not be immunized with the COVID-19 vaccine, as the risk of harm and medical injury to the patient outweigh the benefits.”
She also indicated she had contracted COVID-19 in November 2020 and that she still had antibodies, confirmed by a test conducted internally by Lilly for a study it was running on the previously infected.
The company’s HR department denied Clark’s request, while informing her that there were many ways for her to receive vaccination should she so decide.
In the rejection email sent to Clark, Lilly noted: “This decision was made utilizing the most up to date CDC definition of true medical contraindications to COVID vaccination … with this evidence-based guidance, there are very few scenarios that meet the criteria for medical accommodation.”
Several ex-employees told RCI they had heard Lilly granted few if any medical accommodation requests. Lilly did not respond to questions from RCI aimed at verifying this claim, nor any other questions concerning accommodations.
Like Mandy Van Gorp, Clark said Lilly denied her subsequent request for a religious exemption on the ground that she had missed the application deadline. Internal documents provided to RCI do not reference any appeals process for those seeking leniency from the mandate, and they say nothing about whether an employee could have applied for both a medical and religious accommodation up front.
With her requests rejected, and having refused to get vaccinated, Clark was fired for “misconduct — insubordination.”
Salespeople seeking an exemption from Lilly faced other challenges. Scott, a nearly 20-year Lilly veteran in his mid-50s who asked that his last name not be used because he is looking for a new job, sought a religious exemption based on his long-time opposition to abortion and the fact that cells believed to be from aborted fetuses were used in the testing or development of the COVID-19 vaccines. He wrote a six-page letter detailing his objection, and included a letter from his pastor. He also provided proof that he had COVID-19 antibodies from a previous infection.
To Scott’s surprise, and to the surprise of colleagues who received the same demand, Lilly asked him to send “proof” that fetal cells had been used in vaccine development, including documentation of which fetal cell lines were used. The company also asked other follow-up questions to which several employees with whom RCI spoke objected.
One such email from Lilly HR obtained by RCI required self-identified Catholics to explain why they opposed official Church policy.
If Catholic, please ensure the documentation addresses the determination of the Vatican’s doctrinal office (the Congregation for the Doctrine of the Faith) that it is morally acceptable for Catholics to receive COVID-19 vaccines.
Ohio native Amy Schultz, a sales rep whose religious exemption from getting vaccinated was approved, says that there was another issue at play. She claims the accommodation process itself “wasn’t handled consistently,” noting that “some people were asked for a letter from their pastor. I wasn’t.”

Scott was granted a “temporary religious accommodation.” Then Lilly threw him several curve balls. First, Lilly HR told him in an email that due to the in-person nature of sales, management …
… has determined this accommodation creates an undue hardship for the company and the customers we serve. At this point, you may choose to post for a remote-based role … If you are unable to secure a different non-customer facing position or should you choose not to receive the COVID-19 vaccine by November 15th, you will be terminated from the company.
The non-customer-facing roles to which Lilly directed Scott were for scientists, engineers, and office staff, and many of those, he claims, were based at corporate headquarters in Indianapolis, far from his home out west. Despite having worked at the company for nearly 20 years, he would be treated like any other job applicant. Scott said he applied for six positions – four of which would require him to report to headquarters, and all of which would have resulted in significant pay cuts – and failed to receive an interview for any of them.
Lilly further pressured him by strictly enforcing the terms of a relocation agreement to which Scott had become party two years before. It stipulated that Scott remain employed by Lilly through November 18, 2021 or else be liable for $43,000 in costs the company had incurred to move him and his family. Due to his vaccine-related circumstance, Scott would be terminated two days shy of that date and therefore be on the hook for those moving costs.
Lilly HR offered a solution, but one that Scott felt left him little choice: Accept the severance plan offered to people like him who couldn’t find an acceptable alternative job at Lilly, and the company would waive his repayment obligations. With three kids in college, and the $43,000 bill coming his way, Scott signed the severance agreement and applied for unemployment as he sought a new job.
But Lilly was not done with Scott. The state unemployment office rejected his claim for benefits, asserting, “You were discharged from this [Lilly] employment for violation of company policy.” Scott appealed and received another letter stating he was ineligible because he had quit the company. He appealed again, provided all relevant severance documentation, and explained in a hearing before an unemployment court exactly what had transpired. He asked the Department of Labor representative, “What did Lilly specifically tell you about my employment?” Scott claims that the representative grew dead quiet, then said, “They checked the box that you quit.” Ultimately, the Labor Department approved his benefits.
Other former Lilly sales representatives also reported problems in obtaining unemployment compensation. Two residing in the same state provided letters to RCI from its unemployment office indicating that they were initially denied benefits due to “voluntary leaving” – what they saw as clearly an inaccurate account of their separation from Lilly. One of the two ultimately received benefits, the other did not.
Several former employees left the company over its mandate citing safety concerns with the vaccine rooted in their professional experience. One expressed concern over “adverse events.” By late 2021, he said, he had been observing the Vaccine Adverse Event Reporting System co-managed by the CDC and the Food and Drug Administration and “watching and noticing the staggering amount of AE’s [adverse events] reported from seizures, death, myocarditis. I told my manager, ‘If this was our product and I was seeing these kind of reports, we’d be pulled off the shelves immediately.’”
As of Aug. 12, 2021, the date of the Lilly mandate announcement, the vaccine reporting system had collected over half a million reports. It presents users accessing the data with a disclaimer noting its limitations, including that adverse events may not be attributable to vaccines; that the reports themselves may be incomplete, inaccurate, or otherwise flawed; and that since they are voluntary, they could be subject to biases.
The former salesperson was particularly sensitive to this issue given his position as a pharmaceutical rep, a role in which he says he was obligated to report any and all adverse events encountered in connection with any Lilly drug.
Schultz added: “Where there is a risk, there should be a choice and clearly there is a lot of risk here with this thing and Lilly didn’t care, [about] any of our personal beliefs, it didn’t matter. It’s all about money. It’s all about control.”
Several other sales representatives expressed reservations about taking a vaccine they felt had been hastily brought to market. Van Gorp said, “It’s kind of an oxymoron to be fired for not taking a vaccine that doesn’t have enough information and enough safety data for me to take it myself and give it to my children.”
Another former rep, Amber Nikolai, a military veteran, made a similar point:
Being new to pharma I only knew the training that they had put me through and they put us through so much training that is saturated in understanding the clinical trials … we really had to understand every in and out of the clinical trial and the product info sheet and we had to make sure we could answer every question to be able to help that doc to identify appropriate patients for that medication and to feel good that it had been thoroughly tested and [to] gain their confidence … When this [Lilly imposing its mandate] started happening I thought this is exact opposite of what you teach us. An experimental therapy?
Those interviewed who expressed concerns over risks associated with the vaccines, and their rapid development and launch, cited varying reasons for their caution: Some referenced growing adverse-event figures. Others noted Israeli studies on waning vaccine efficacy. Still others leaned on anecdotal evidence from friends and family they claim had suffered adverse reactions to the jabs. How those issues should be weighed against a once-in-a-century pandemic went unstated.
Lilly, in a Q&A document obtained by RCI that was delivered to employees in conjunction with its Aug. 12 vaccine mandate announcement, noted:
From a safety standpoint, more than 4 billion doses of coronavirus vaccines have been administered worldwide. In the U.S. alone, more than 347 million doses have been administered. The Centers for Disease Control and Prevention (CDC) launched the largest vaccine adverse event tracking system in history for the COVID-19 vaccines. Since the vaccines became available, reported severe side effects have been uncommon. As a science-based company, we have thoroughly reviewed all the data and options available to us. We believe this decision helps keep our employees, families and customers safe and healthy, and ensures we can continue making life-saving medicines for people around the world.
Lilly also referenced a sense of urgency to get employees vaccinated, rooted in science: “We’re making this decision ahead of the anticipated full approval of the vaccines by the FDA, which is imminent, because we believe every day counts. Science tells us that the current vaccines are effective in slowing transmission and reducing severe illness and deaths.”
For the salespeople who left the company, another twist awaited them: The day the mandate went into effect, Lilly reported to employees that a small portion of unvaccinated customer-facing employees would be given permission to work virtually, acknowledging that “experts now believe that fully vaccinated individuals with COVID can transmit the virus at the same rate as those who are not vaccinated.”
On that date, Lilly HR reported in an email to employees that “99% of U.S. employees met the vaccination requirement or have an approved medical or religious accommodation in place.”
Today, COVID-19 restrictions are easing across the country, and some employers are following suit. United Airlines, for example, is allowing unvaccinated employees who had been out on approved accommodation requests to return to the office.
For his part, Scott said he would not go back to Lilly if given the opportunity. He wrote a letter to his colleagues on his final day: “An employer that thinks they have the right to tell you what to put in your body with no liability is not a company that I want to work for.”
Nikolai, who was granted a religious accommodation but did not pursue a non-customer-facing role and was separated from Lilly, turned down a severance agreement. “For me, my religion can’t be bought. My freedom can’t be bought. There have to be some people that are willing to stand.”
She is now pursuing legal action against the company.
“Am I going to win against a multibillion dollar pharma company who is insulated five ways from Sunday? No, their pockets are deep. But someone’s gotta stand and say this is wrong, if we don’t try, where does it end?”
Reprinted from RealClearInvestigations
Join the conversation:

Published under a Creative Commons Attribution 4.0 International License
For reprints, please set the canonical link back to the original Brownstone Institute Article and Author.









